Human epidermal growth factor receptor 2 (HER2) in colorectal carcinoma
ERBB2 gene amplification and HER2 protein overexpression occur in 2–5% of colorectal carcinomas and may predict response to targeted anti-HER2 monoclonal antibody therapies in patients with advanced disease. This article discusses the utility of next-generation sequencing and HER2 immunohistochemistry as biomarker assays for optimal selection of patients who may benefit from HER2 inhibitors.
by Dr Odise Cenaj
Introduction
Colorectal carcinoma, a malignant tumour arising from the epithelial lining of the large intestine, remains a leading cause of cancer-related deaths worldwide. Each year, in the United States of America alone, approximately 150 000 patients are diagnosed with colorectal cancer and approximately 50 000 succumb to this disease [1]. The increased adoption of screening and preventative measures, such as colonoscopy, sigmoidoscopy and fecal occult blood testing, has improved mortality by detecting early stage cancers in asymptomatic patients. However, the vast majority of newly diagnosed patients presents with locally advanced cancer and 1 in 5 patients will have metastatic disease at presentation [2].
Endoscopic or surgical resection alone can be curative for early stage tumours (stages I and II), but chemotherapy is indicated for patients with locally advanced or metastatic cancer (stages III and IV). Conventional non-targeted cytotoxic chemotherapeutic agents (such as 5-fluorouracil, capecitabine, irinotecan, and oxaliplatin, whose mechanism of action is inhibition of DNA synthesis and replication), remain the mainstay treatment, recent discovery of targeted molecular therapies with humanized monoclonal antibodies – bevacizumab targeting vascular endothelial growth factor receptor, cetuximab and panitumumab targeting epidermal growth factor receptor (EGFR) – have shown significant improvement in overall and progression-free survival [3–5].
Targeted molecular therapies in colorectal cancer
A targeted molecular therapy is a pharmacologic agent that is designed to bind with high specificity to a particular cellular molecule whose aberrant structure or function is uniquely present in a particular type of cancer cell, but absent in non-cancer cells. This aberrant molecule typically drives cellular signalling pathways that lead to the expression of genes that promote cancer cell proliferation, survival, inhibition of apoptosis, and increased potential for invasion and metastasis. Binding of the pharmacologic agent to the target molecule blocks signalling via these pathways and leads to cancer cell death.
An exemplary molecular target is human epidermal growth factor receptor 2 (HER2), a transmembrane receptor tyrosine kinase encoded by the Erb-b2 receptor tyrosine kinase 2 (ERBB2) gene on chromosome 17 [6]. ERBB2 gene amplification leads to HER2 protein overexpression and accumulation on the cellular membrane. This accumulation results in higher rates of receptor dimerization, which in turn triggers activation of phosphoinositide-3-kinase (PI3K)/protein kinase B (Akt) and mitogen-activated protein kinase (MAPK) signalling pathways, responsible for tumour proliferation and survival [7].
A common and effective strategy for expanding the scope of molecular targeted therapies is choosing existing agents with an established efficacy on a particular type of cancer and investigating its role in another type of cancer. An excellent example of such a success story is the role of HER2 in colorectal carcinoma. ERBB2 amplification and/or HER2 overexpression is a central molecular target in breast and gastroesophageal cancers, and monoclonal antibodies against HER2 have improved outcomes in these patients [8, 9]. As a result, testing and detection of ERBB2 amplification by fluorescence in situ hybridization (FISH) or chromogenic in situ hybridization (CISH) and HER2 protein overexpression via immunohistochemistry using validated scoring systems is now considered standard care in anatomic pathology practice [10, 11].
Role of HER2 in colorectal cancer
A seminal study by the Cancer Genome Atlas Network, which used whole genome sequencing for the molecular characterization of human colorectal cancer, found that the ERBB2 locus was recurrently amplified in 4 % of tumours [12]. Other studies that used FISH and immunohistochemistry methods have reported similar rates ranging from 2 % to 5 % [13, 14]. Although this prevalence is relatively lower than that seen in breast cancer, this percentage still represents a sizable number of patients with colorectal cancer and the therapeutic potential of HER2 inhibitors is particularly promising for those with metastatic disease who have failed prior rounds of conventional chemotherapy or who have developed resistance to inhibitors against EGFR, another major molecular target in colorectal carcinoma. More recently, a phase II clinical trial of patients with KRAS wild-type, cetuximab-resistant, metastatic colorectal carcinoma showed that the presence of ERBB2 amplification and/or HER2 protein overexpression predicted response to combined targeted therapy with HER2 inhibitors trastuzumab and lapatinib with an overall response rate of 30 % [HER2 Amplification for Colorectal Cancer Enhanced Stratification (HERACLES) trial] [15]. As the ERBB2 gene is located on chromosome 17, ERBB2 copy number is reported as a ratio to a chromosome enumeration probe 17 (CEP17). Patients were selected and included in this trial if their tumours were positive for ERBB2 amplification by FISH (defined as an ERBB2/CEP17 ratio of 2 or more by FISH) and/or HER2 overexpression by immunohistochemistry (defined as intense membranous staining in 50 % or more of tumour cells). These criteria for positivity were developed by the HERACLES investigators by adapting existing scoring systems of HER2 expression and ERBB2 amplification in breast and gastric cancer [10, 11]. These HER2 immunohistochemistry scoring systems combine the intensity of membranous staining with the percentage of tumour cells staining, and the result is expressed on a semi-quantitative scale from 0 to 3+.
Despite these advances, colorectal carcinomas are not routinely screened for ERBB2 amplification or HER2 protein overexpression in daily clinical practice for several reasons. The cost-to-benefit ratio of FISH and/or immunohistochemistry screening of every colorectal cancer case remains prohibitive in most pathology laboratories around the world, particularly when combined with the rarity of this event. In addition, in a significant percentage of patients, tissue available for testing is limited to biopsy material, and testing for the presence of other more clinically established oncogenic biomarkers [such as B-Raf proto-oncogene, serine/threonine kinase (BRAF) and KRAS proto-oncogene, GTPase (KRAS)] takes precedence over HER2 status. This obstacle can be circumvented by deploying assays that can investigate multiple targets at once.
Next-generation sequencing
Such an example is next-generation sequencing (NGS), an assay that is increasingly used for the simultaneous detection of multiple prognostic and predictive markers in cancer patients [16]. In brief, NGS is a DNA sequencing assay where millions of fragments of genomic DNA are sequenced in parallel. Tumour DNA is isolated via manual macrodissection of unstained sections of formalin-fixed paraffin-embedded tissue blocks. Tumour DNA percentage is estimated in the pre-analytical phase by an anatomic pathologist using light microscopy on a corresponding hematoxylin and eosin stained slide. The minimum estimated tumour DNA percentage required for most assays for an acceptable analytical sensitivity is set at 20–30 %. Following DNA isolation and parallel sequencing, bioinformatics tools are used to link the nucleotide sequence data of the fragments by mapping them on a reference human genome [17, 18]. ‘Depth of coverage’ is a measure of how many times a single DNA base is sequenced in particular run and is a reflection of the quality of the data. While some assays deploy whole genome or whole exome sequencing, NGS used in molecular oncologic pathology typically targets the exons and introns genes that are known to be associated with human cancer. Aside from small-scale mutations (substitutions, insertions, and deletions), NGS can readily identify large-scale DNA copy number alterations at the chromosome or chromosomal arm level, but also focal amplification events, including those in regions containing the ERBB2 gene. Finally, copy number alterations are detected and called using customized bioinformatics pipelines [16, 18].
NGS versus immunohistochemistry for the detection of ERBB2-amplified colorectal cancer
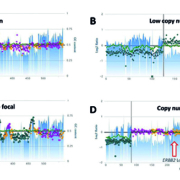
The utility of NGS as a robust and stand-alone assay in detecting ERBB2 amplification was demonstrated in a recent study of breast and gastric cancer, where NGS calls had an overall concordance rate of 98.4 % with combined immunohistochemistry/FISH results [19]. A similarly high concordance between NGS and immunohistochemistry was also shown in colorectal carcinoma [20, 21]. In the study by Cenaj et al., ERBB2 amplification by NGS (defined as 6 copies or more) was correlated with HER2 overexpression by immunohistochemistry (Fig. 1; monoclonal antibody SP3; semi-quantitatively assessed using H-scores) in a cohort of 102 colorectal carcinoma patients that was retrospectively selected to represent a wide range of ERBB2 copy number values: 15 cases with ERBB2 amplification, 10 with low copy number gains at the chromosome or chromosomal arm level, and 77 copy number neutral cases (Fig. 2)[20]. The data suggest that ERBB2 amplification in colorectal carcinoma is a high-level focal event, with estimated copy numbers ranging from 14 to over 100. Furthermore, HER2 expression in colorectal carcinoma appears to follow a bimodal distribution, with all ERBB2-amplified tumours by NGS showing a HER2 immunohistochemistry H-score of 105 or more and non-amplified tumours clustering around zero to weak HER2 immunostaining.
HER2 immunohistochemistry has several limitations that are overcome by NGS: the need for stringent tissue fixation times, intraobserver and interobserver variability in scoring, and differences in performance characteristics (assay sensitivity and specificity) which can vary widely depending on the platform (Ventana versus HercepTest) and antibody clone (4B5 versus SP3) used for HER2 immunohistochemistry. These limitations obviate the need for inclusion of confirmatory FISH testing. In our experience, when setting the HERACLES diagnostic criteria for ERBB2/HER2 positivity as threshold, not only does NGS accurately identify all cases positive by immunohistochemistry, but it also detects cases with ERBB2 amplification that would have otherwise been considered equivocal or negative by immunohistochemistry by these same criteria. This observation suggests that NGS may be more sensitive than immunohistochemistry in detecting patients with colorectal cancer who could benefit from HER2 inhibitor therapy.
On the other hand, the sensitivity of detection of copy number alterations by NGS is dependent on tumour DNA content, among other technical factors [19], and samples with poor DNA quality or low tumour fraction may lead to false negative results. In addition, a significant portion of colorectal cancers displays significant spatial heterogeneity of HER2 expression by immunohistochemistry, including tumours that demonstrate high ERBB2 amplification by NGS. This finding suggests that not all ERBB2 amplification events lead to the same level of HER2 protein expression and that the association between gene amplification and protein overexpression may be more complex that initially anticipated. Since the efficacy of HER2 inhibitors may depend on the distribution of HER2 protein on the cell surface and other mechanisms that affect receptor stability and degradation via endocytosis [22], HER2 immunohistochemistry will continue to provide valuable information by direct demonstration of HER2 expression at the cellular level and should continue to complement NGS testing as a biomarker of response to HER2 inhibitors.
Concluding remarks
NGS accurately detects ERBB2 amplification in colorectal carcinoma and shows high concordance with HER2 positivity by immunohistochemistry. Although NGS can be used as a stand-alone assay for the simultaneous interrogation of multiple cancer biomarkers including ERBB2 amplification, HER2 immunohistochemistry is still needed to demonstrate heterogeneity at the protein level. Screening for ERBB2 amplification by NGS in combination with complementary evaluation of HER2 expression by immunohistochemistry may provide optimal prediction of response to HER2 inhibitors in patients with colorectal carcinoma.
Figure 1. Copy number variation plots of chromosome 17 from next-generation sequencing data in four patients with colorectal carcinoma. The dots denote the log2 ratio of the target coverage of the tumour sample to a panel of normal non-neoplastic tissues. The alternating gold and magenta colours highlight individual gene boundaries. Relative GC nucleotide content is highlighted by blue tracings. (a) Amplification of the ERBB2 locus in 17q. (b) Low copy number gain at the chromosomal arm level, including the ERBB2 locus. (c) Low focal copy number gain at the ERBB2 locus. (d) Neutral copy number for 17q with concurrent loss of chromosomal arm 17p. (Reproduced from Cenaj O, Ligon AH, Hornick JL, Sholl LM. Detection of ERBB2 amplification by next-generation sequencing predicts HER2 expression in colorectal carcinoma. Am J Clin Pathol 2019; 152(1): 97–108, by permission of Oxford University Press.)
Figure 2. Range of HER2 protein expression patterns in colorectal carcinoma as detected by immunohistochemistry. (a) Strong membranous staining (scored as 3+). (b, c) Weak to moderate membranous staining (1 to 2+). (d) Negative (0) membranous staining. (Reproduced from Cenaj O, Ligon AH, Hornick JL, Sholl LM. Detection of ERBB2 amplification by next-generation sequencing predicts HER2 expression in colorectal carcinoma. Am J Clin Pathol 2019; 152(1): 97–108, by permission of Oxford University Press.)
References
1. Siegel RL, et al. Cancer statistics, 2019. CA Cancer J Clin 2019; 69(1): 7–34.
2. Siegel RL, et al. Cancer statistics, 2016. CA Cancer J Clin 2016; 66(1): 7–30.
3. André T, et al. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J Clin Oncol 2009; 27(19): 3109–3116.
4. Karapetis CS, et al. K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N Engl J Med 2008; 359: 1757–1765.
5. Cremolini C, et al. FOLFOXIRI plus bevacizumab versus FOLFIRI plus bevacizumab a first-line treatment of patients with metastatic colorectal cancer: updated overall survival and molecular subgroup analyses of the open-label, phase 3 TRIBE study. Lancet Oncol 2015; 16(13): 1306–1315.
6. Coussens L, et al. Tyrosine kinase receptor with extensive homology to EGF receptor shares chromosomal location with neu oncogene. Science 1985; 230(4730): 1132–1139.
7. Kirouac DC, et al. HER2+ cancer cell dependence on PI3K vs. MAPK signaling axes is determined by expression of EGFR, ERBB3 and CDKN1B. PLoS Comput Biol 2016; 12(4): e1004827.
8. Slamon DJ, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 2001; 344(11): 783–792.
9. Bang YJ, et al; ToGA Trial Investigators. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 2010; 376(9742): 687–697.
10. Hofmann M, et al. Assessment of a HER2 scoring system for gastric cancer: results from a validation study. Histopathology 2008; 52(7): 797–805.
11. Wolff AC, et al. Human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline focused update. Arch Pathol Lab Med 2018; 142(11): 1364–1382.
12. The Cancer Genome Atlas Network. Comprehensive molecular characterization of human colon and rectal cancer. Nature 2012; 487(7407): 330–337.
13. Nathanson DR, et al. HER 2/neu expression and gene amplification in colon cancer. Int J Cancer 2003; 105(6): 796–802.
14. Ooi A, et al. Protein overexpression and gene amplification of HER-2 and EGFR in colorectal cancers: an immunohistochemical and fluorescent in situ hybridization study. Mod Pathol 2004; 17(8): 895–904.
15. Sartore-Bianchi A, et al. Dual-targeted therapy with trastuzumab and lapatinib in treatment-refractory, KRAS codon 12/13 wild-type, HER2-positive metastatic colorectal cancer (HERACLES): a proof-of-concept, multicentre, open-label, phase 2 trial. Lancet Oncol 2016; 17(6): 738–746.
16. Sholl LM, et al. Institutional implementation of clinical tumor profiling on an unselected cancer population. JCI Insight 2016; 1(19): e87062.
17. Behjati S, Tarpey PS. What is next generation sequencing? Arch Dis Child Educ Pract Ed 2013; 98(6): 236–238.
18. Garcia EP, et al. Validation of OncoPanel: a targeted next-generation sequencing assay for the detection of somatic variants in cancer. Arch Pathol Lab Med 2017; 141(6): 751–758.
19. Ross DS, et al. Next-generation assessment of human epidermal growth factor receptor 2 (ERBB2) amplification status: clinical validation in the context of a hybrid capture-based, comprehensive solid tumor genomic profiling assay. J Mol Diagn 2017; 19(2): 244–254.
20. Cenaj O, et al. Detection of ERBB2 amplification by next-generation sequencing predicts HER2 expression in colorectal carcinoma. Am J Clin Pathol 2019; 152(1): 97–108.
21. Shimada Y, et al. Utility of comprehensive genomic sequencing for detecting HER2-positive colorectal cancer. Hum Pathol 2017; 66: 1–9.
22. Pereira PMR, et al. Caveolin-1 mediates cellular distribution of HER2 and affects trastuzumab binding and therapeutic efficacy. Nat Commun 2018; 9(1): 5137.
The author
Odise Cenaj MD
Department of Pathology, New York University Langone Health, New York, NY 10016, USA
E-mail: odise.cenaj@nyulangone.org